Abstract
Background: Allogeneic hematopoietic cell transplantation (allo-HCT) management has evolved, with the emergence of molecular detection assays and therapies to treat post-HCT complications such as graft-versus-host disease (GVHD). However, this approach could increase the economic burden on healthcare systems by only managing complications that occur after the initial transplant. Instead, alternative therapies involving novel engineered donor allografts to replace standard allo-HCT to prevent late complications may potentially improve outcomes and address a greater unmet need. Objective: To estimate (1) the average total lifetime medical costs of an allo-HCT and (2) to estimate the net monetary savings and value associated with reducing late complications.
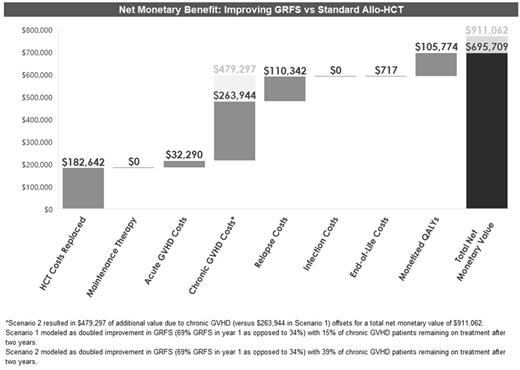
Methods: A short-term decision tree (100-day) and a long-term semi-Markov partitioned survival model were developed that estimated the average per-patient lifetime cost and expected quality-adjusted life years (QALYs) for an allo-HCT patient from a United States healthcare system perspective. The allo-HCT patient population represented an average 53-year-old patient who was undergoing an allo-HCT to treat the three most common indications for allo-HCT: acute myeloid leukemia (AML), acute lymphoblastic leukemia (ALL), or myelodysplastic syndromes (MDS), distributed as 51%, 21%, and 28%, respectively. Key model clinical inputs included: overall survival, GVHD-free relapse-free survival (GRFS), incidence of both acute and chronic GVHD, relapse of the primary disease, and infections. Following the first 100 days, the patient cohort was assigned to 3 mutually exclusive health states in a semi-Markov partitioned survival model for the remainder of their lifetime: (1) GRFS, (2) progressed and/or GVHD, and (3) death. The projected time spent in each health state was derived from data provided by CIBMTR. Costs pertaining to allo-HCT and associated complications were obtained from the published literature based on medical claims analyses of payments (paid amounts) and were converted into costs using the American Hospital Association (AHA) commercial payment-to-cost ratio. Maintenance therapy costs for FLT3-ITD positive AML and Ph+ ALL cases were also included and were based on published literature, expert opinion, and prescribing information. The key outcomes were direct medical costs and expected QALYs. Cost results were reported as a range based on varying the percent of chronic GVHD patients that remained on treatment after two years (15% or 39%) as derived from the published literature. Scenario analyses were conducted to estimate the potential clinical and economic value that may be realized by improving GRFS outcomes. The scenario analyses to address some of the unmet need were modeled as a doubled improvement in GRFS (69% GRFS in year 1 as opposed to 34%) to estimate changes in allo-HCT-associated costs and survival. A willingness-to-pay threshold of $150,000 was used to monetize the QALYs gained as a result of improving GRFS outcomes.
Results: Over a lifetime, the average per-patient medical cost of allo-HCT was estimated to range from $942,373 to $1,247,917. Of those total costs, 23% to 30% were incurred within the 1st 100 days and 42% to 56% within the 1st year. Of the medical costs estimated, the majority of the costs were for chronic GVHD treatment (37% to 53%) followed by the allo-HCT procedure (15% to 19%). Expected lifetime QALYs of an allo-HCT patient were estimated to be 4.7. In the scenario analyses, the unmet need that could be addressed by improving GRFS outcomes (via the medical cost offsets and QALYs gained) was associated with a net monetary value that ranged from $695,709 to $911,062. Of the estimated net monetary value, 85% to 88% was driven by direct medical cost savings while the remaining 12% to 15% was due to value of the gain in expected QALYs.
Conclusions: The current per-patient cost of allo-HCT carries a substantial medical cost burden often exceeding $1M over the lifetime of the patient. Innovative research efforts focused on disease control and reduction or elimination of late complications altogether, particularly cGVHD, may provide the greatest value in improved patient outcomes and reduced healthcare spending.
Disclosures
Maziarz:ASTCT: Membership on an entity's Board of Directors or advisory committees; Orca Bio: Other: Support for research analysis and for medical writing; Allovir: Other: Support for research on Allo HCT costs of care of infectious related complications; CRISPR Therapeutics: Consultancy, Honoraria; Novartis: Other: Support for research on CART. Devine:Orca Bio: Consultancy, Other: Payment to NMDP for consulting. Garrison:BluePath Solutions, Inc.: Consultancy, Other: Fees for independent health economics consulting. Agodoa:Orca Bio: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. Badaracco:BluePath Solutions, Inc.: Current Employment; Orca Bio: Consultancy, Other: receives consulting fees from Orca Bio outside the submitted work . Gitlin:BluePath Solutions, Inc.: Current Employment; Orca Bio: Consultancy, Other: receives consulting fees from Orca Bio outside the submitted work . Perales:DSMB: Other; Sellas Life Sciences: Consultancy; Astellas: Honoraria; Abbvie: Honoraria; Medigene: Consultancy; Cidara Therapeutics: Consultancy; Orca Bio: Consultancy; Novartis: Honoraria; Vor Biopharma: Honoraria; Nektar Therapeutics: Consultancy, Honoraria; Kite, a Gilead Company: Honoraria, Research Funding; Servier: Consultancy; Merck: Consultancy; VectivBio AG: Honoraria; Omeros: Consultancy; Miltenyi Biotec: Consultancy, Honoraria; Takeda: Honoraria; Bellicum: Honoraria; Celgene: Honoraria; Karyopharm: Honoraria; MorphoSys: Consultancy, Honoraria; Incyte: Honoraria, Research Funding; Bristol-Mysers Squibb: Honoraria.
OffLabel Disclosure:
Maintenance sorafenib after allo-HCT for FLT3-ITD positive AML and maintenance imatinib after allo-HCT for Ph+ ALL
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal